Our Mission
Trusted in the operating room.
Our History
Atlanta Healthcare Services, Inc. (AHS) is a privately held U.S. medical device company specializing in the design and development of ophthalmic and oculoplastic surgical drapes and fluid control solutions. Through its OcuDrape® brand, AHS delivers purpose-built products that enhance surgical efficiency, improve fluid management, and maintain a reliable sterile field in operating rooms worldwide.
With decades of experience in surgical drape innovation, AHS was established during the early transition from reusable textiles to single-use, high-barrier surgical systems—helping to advance infection control standards and operating room efficiency. Early in its evolution, the company worked closely with hospitals and surgeons to develop customized surgical solutions, contributing to foundational innovations in procedure-specific product design.
Building on this expertise, AHS strategically refined its focus to concentrate on specialty surgical drapes—particularly within ophthalmology and oculoplastic procedures—where precision, fluid control, and consistency are critical. This focused approach has enabled the company to develop a highly specialized portfolio tailored to the unique demands of eye surgery.
Today, OcuDrape® products are engineered using premium materials and manufactured through FDA-registered and ISO-certified partners to ensure consistent quality and performance. The product line includes a wide range of drape configurations designed to optimize surgical access, control fluid runoff, and streamline operating room setup.
AHS maintains long-standing relationships across the ophthalmic surgical ecosystem, with products incorporated into custom procedure packs and used by leading healthcare providers. Known for its combination of design expertise, manufacturing flexibility, and cost-effective solutions, AHS continues to deliver high-performance products that meet the evolving needs of surgeons and healthcare systems.
From pioneering surgical drape design to advancing specialty solutions, AHS continues to set the standard in the ophthalmic industry.
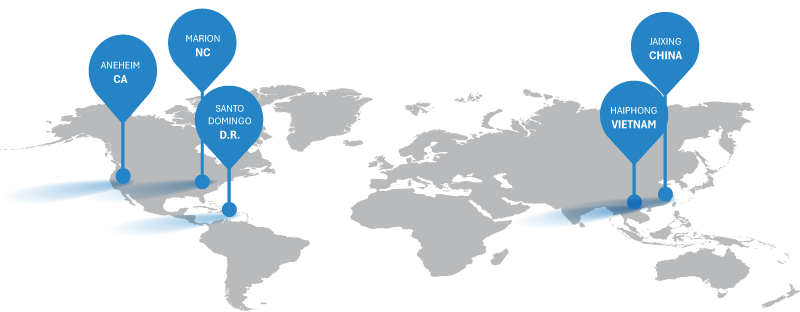
Global Manufacturing Network & Quality Standards
AHS leverages a globally diversified network of manufacturing partners across North America, Asia, and the Caribbean—each selected for their expertise in medical device manufacturing. All facilities are ISO 13485 certified and FDA registered, ensuring consistent quality and regulatory compliance. With full control over product design and material specifications, AHS delivers product consistency across all sites while maintaining the flexibility to shift production as needed. The result is a resilient, scalable supply chain that ensures reliable product availability for customers worldwide.